Recombinant Human Tumor Necrosis Factor-α/TNF-α High Active Mutant
| Product name: | Recombinant Human Tumor Necrosis Factor-α/TNF-α High Active Mutant |
| Source: | E.coli |
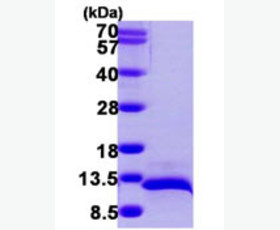
| Purity: | Greater than 95% as determined by reducing SDS-PAGE. |
| Buffer Formulation: | Lyophilized from a 0.2 μm filtered solution of 20mM PB, 150mM NaCl, pH 7.4. |
| Applications: | Applications:SDS-PAGE; WB; ELISA; IP. |
| Storage: | Avoid repeated freeze/thaw cycles. Store at 2-8 oC for one month. Aliquot and store at -80 oC for 12 months. |
| UOM: | 100ug/50ug/200ug/1mg/1g |
| Source | E.coli |
| Description | Recombinant Human Tumor Necrosis Factor alpha is produced by our E.coli expression system and the target gene encoding is expressed. |
| Names | Tumor Necrosis Factor, Cachectin, TNF-Alpha, Tumor Necrosis Factor Ligand Superfamily Member 2, TNF-a, TNF, TNFA, TNFSF2 |
| Accession # | P01375 |
| Formulation | Lyophilized from a 0.2 μm filtered solution of 20mM PB, 150mM NaCl, pH 7.4. |
| Shipping |
The product is shipped at ambient temperature. |
| Reconstitution |
Always centrifuge tubes before opening. Do not mix by vortex or pipetting. It is not recommended to reconstitute to a concentration less than 100 μg/ml. Dissolve the lyophilized protein in ddH2O. Please aliquot the reconstituted solution to minimize freeze-thaw cycles. |
| Storage |
Lyophilized protein should be stored at < -20°C, though stable at room temperature for 3 weeks. Reconstituted protein solution can be stored at 4-7°C for 2-7 days. Aliquots of reconstituted samples are stable at < -20°C for 3 months. |
| Biological Activity |
ED50 is less than 0.01 ng/ml. Specific Activity of 1.0 x 10^8 IU/mg. |
| Purity |
Greater than 95% as determined by reducing SDS-PAGE. |
| Endotoxin | Less than 0.1 ng/µg (1 IEU/µg) as determined by LAL test. |
| Background | Tumor Necrosis Factor-α (TNF-α) is secreted by macrophages, monocytes, neutrophils, T-cells, and NK-cells following stimulation by bacterial LPS. Cells expressing CD4 secrete TNF-α while cells that express CD8 secrete little or no TNF-α. Synthesis of TNF-α can be induced by many different stimuli including interferons, IL2, and GM-CSF. The clinical use of the potent anti-tumor activity of TNF-α has been limited by the proinflammatory side effects such as fever, dose-limiting hypotension, hepatotoxicity, intravascular thrombosis, and hemorrhage. Designing clinically applicable TNF-α mutants with low systemic toxicity has been of intense pharmacological interest. Human TNF-α that binds to murine TNF-R55 but not murine TNF-R7, exhibits retained anti-tumor activity and reduced systemic toxicity in mice compared with murine TNF-α, which binds to both murine TNF receptors. Based on these results, many TNF-α mutants that selectively bind to TNF-R55 have been designed. These mutants displayed cytotoxic activities on tumor cell lines in vitro and have exhibited lower systemic toxicity in vivo. Recombinant Human TNF-α High Active Mutant differs from the wild-type by amino acid subsitution of amino acids 1-7 with Arg8, Lys9, Arg10 and Phe157. This mutant form has been shown to have increased activity with less inflammatory side effects in vivo. |
| References |
Wu Y,et al.DDIT3 overexpression increases odontoblastic potential of human dental pulp cells PMID:24738922 http://www.ncbi.nlm.nih.gov/pubmed/24738922 |